Allergy Antihistamine (diphenhydramine hydrochloride 25 mg) Dailymed
Generic: diphenhydramine hydrochloride is used for the treatment of Anxiety Disorders Infant, Newborn Infant, Premature Sleep Initiation and Maintenance Disorders Lactation Motion Sickness Nausea Rhinitis, Allergic, Perennial Sneezing Status Asthmaticus Urticaria
IMPRINT: T 061
SHAPE: capsule
COLOR: pink
All Imprints
diphenhydramine hydrochloride 25 mg - t 061 capsule pink
diphenhydramine hydrochloride 25 mg - t 061 oval pink
Go PRO for all pill images
Otc - Active Ingredient Section
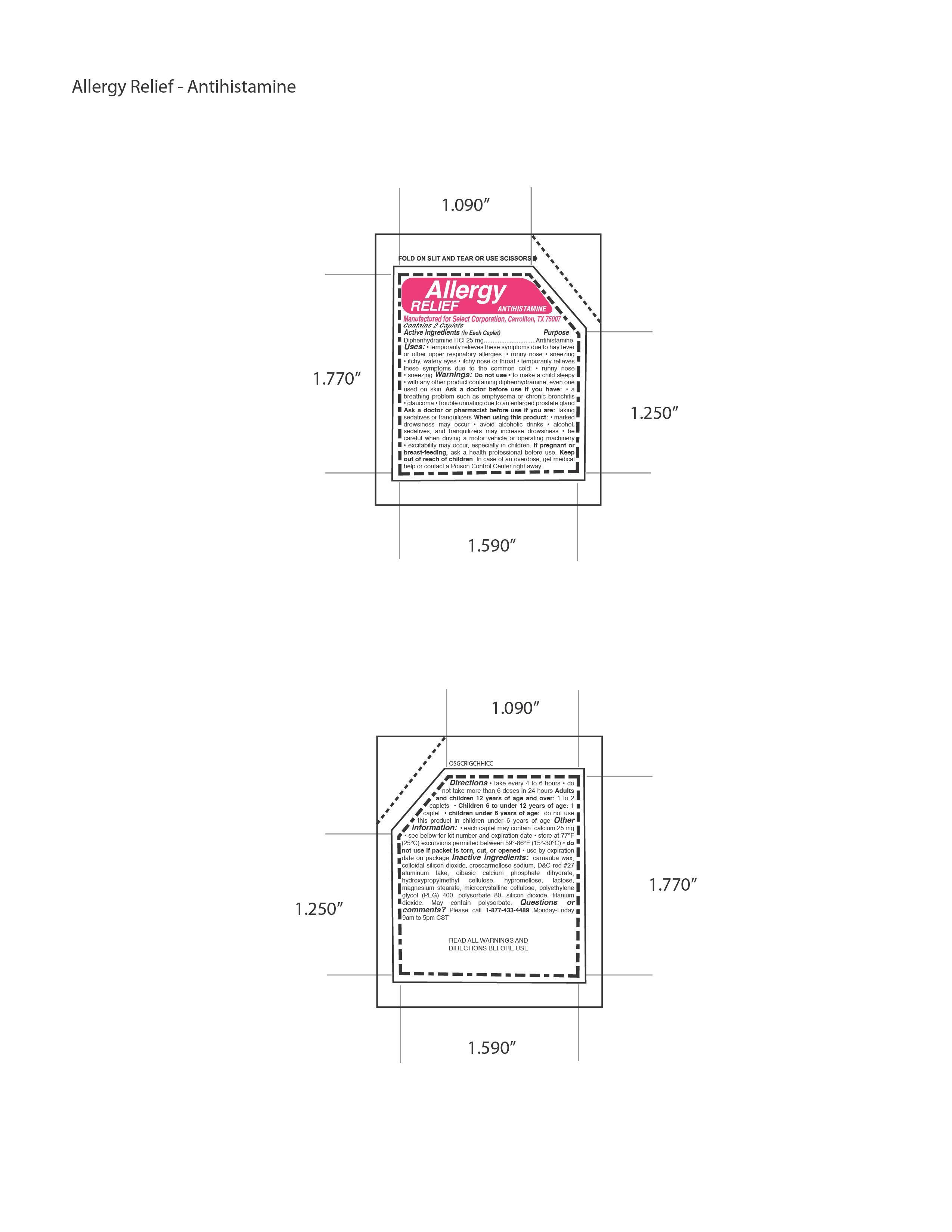
Active Ingredients
Diphenhydramine HCl 25 mg
Otc - Purpose Section
Antihistamine
Dosage & Administration Section
Directions • take every 4 to 6 hours • donot take more than 6 doses in 24 hours Adultsand children 12 years of age and over: 1 to 2caplets • Children 6 to under 12 years of age: 1caplet • children under 6 years of age: do not usethis product in children under 6 years of age
Indications & Usage Section
Uses: • temporarily relieves these symptoms due to hay feveror other upper respiratory allergies: • runny nose • sneezing• itchy, watery eyes • itchy nose or throat • temporarily relievesthese symptoms due to the common cold: • runny nose• sneezing
Warnings Section
Warnings: Do not use • to make a child sleepy• with any other product containing diphenhydramine, even oneused on skin Ask a doctor before use if you have: • abreathing problem such as emphysema or chronic bronchitis• glaucoma • trouble urinating due to an enlarged prostate glandAsk a doctor or pharmacist before use if you are: takingsedatives or tranquilizers When using this product: • markeddrowsiness may occur • avoid alcoholic drinks • alcohol,sedatives, and tranquilizers may increase drowsiness • becareful when driving a motor vehicle or operating machinery• excitability may occur, especially in children.
Otc - Pregnancy Or Breast Feeding Section
If pregnant orbreast-feeding, ask a health professional before use.
Otc - Keep Out Of Reach Of Children Section
KEEP OUT OF REACH OF CHILDREN.
Inactive Ingredient Section
Inactive ingredients: carnauba wax,colloidal silicon dioxide, croscarmellose sodium, DC red 27aluminum lake, dibasic calcium phosphate dihydrate,hydroxypropylmethyl cellulose, hypromellose, lactose,magnesium stearate, microcrystalline cellulose, polyethyleneglycol (PEG) 400, polysorbate 80, silicon dioxide, titaniumdioxide
Package Label.principal Display Panel
MM1
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site


