Esomeprazole Magnesium (esomeprazole magnesium 20 mg) Dailymed
Generic: esomeprazole magnesium is used for the treatment of Duodenal Ulcer Esophagitis Gastroesophageal Reflux Heartburn Stomach Ulcer Zollinger-Ellison Syndrome Helicobacter Infections
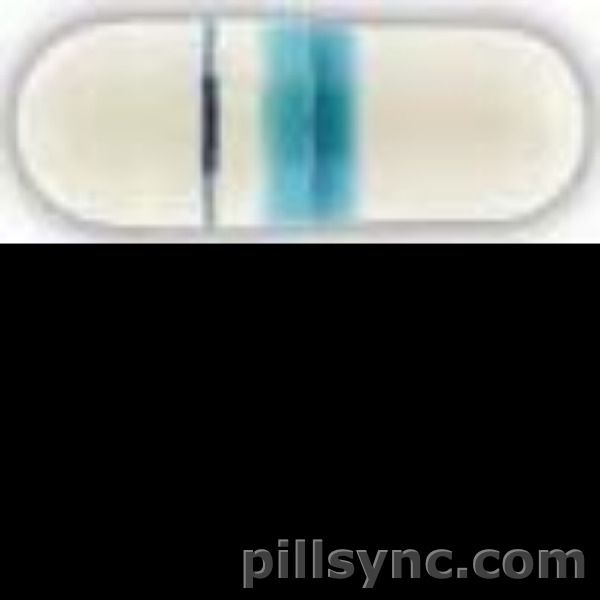
IMPRINT: AMNEAL1050
SHAPE: capsule
COLOR: white
All Imprints
esomeprazole magnesium capsule, delayed release - amneal 1050 capsule white
esomeprazole 20 mg delayed release oral capsule - amneal1050 capsule white
Go PRO for all pill images
Drug Facts
Active ingredient
(in each capsule)
Purpose
Esomeprazole 20 mg
Acid reducer
(*Each delayed-release capsule corresponds to 22.3 mg esomeprazole magnesium trihydrate)
Uses
- treats frequent heartburn (occurs 2 or more days a week)
- not intended for immediate relief of heartburn; this drug may take 1 to 4 days for full effect
Warnings
Allergy alert:
- Do not use if you are allergic to esomeprazole
- Esomeprazole may cause severe skin reactions. Symptoms may include:
- skin reddening
- bulers
- rash
If an allergic reaction occurs, stop use and seek medical help right away.
Do not use if you have
- trouble or pain swallowing food, vomiting with blood, or bloody or black stools
- heartburn with lightheadedness, sweating or dizziness
- chest pain or shoulder pain with shortness of breath; sweating; pain spreading to arms, neck or shoulders; or lightheadedness
- frequent chest pain
These may be signs of a serious condition. See your doctor.
Ask a doctor before use if you have
- had heartburn over 3 months. This may be a sign of a more serious condition.
- frequent wheezing, particularly with heartburn
- unexplained weight loss
- nausea or vomiting
- stomach pain
OTC - ASK DOCTOR/PHARMACIST SECTION
Ask a doctor or pharmacist before use if you are
- taking a prescription drug. Acid reducers may interact with certain prescription drugs.
Stop use and ask a doctor if
- your heartburn continues or worsens
- you need to take this product for more than 14 days
- you need to take more than 1 course of treatment every 4 months
- you get diarrhea
- you develop a rash or joint pain
If pregnant or breast-feeding,
 ask a health professional before use.
Keep out of the reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- adults 18 years of age and older
- this product is to be used once a day (every 24 hours), every day for 14 days
- may take 1 to 4 days for full effect
14-Day Course of Treatment
- swallow 1 capsule with a glass of water before eating in the morning
- take every day for 14 days
- do not take more than 1 capsule a day
- swallow whole. Do not crush or chew capsules.
- do not use for more than 14 days unless directed by your doctor
Repeated 14-Day Courses (if needed)
- you may repeat a 14-day course every 4 months
- do not take for more than 14 days or more often than every 4 months unless directed by a doctor
- children under 18 years of age: ask a doctor before use. Heartburn in children may sometimes be caused by a serious condition.
Other Information
- read the directions and warnings before use
- keep the carton. It contains important information.
- store at 20° to 25°C (68° to 77°F)
- Tamper Evident: Do not use if printed safety seal under cap or band around the center of each capsule is broken or missing.
Inactive Ingredients
gelatin, hypromellose, magnesium stearate, methacrylic acid copolymer dispersion type C, mono-and di-glycerides, polysorbate 80, sugar sphere (contains- sucrose, corn starch and purified water), sodium lauryl sulphate, talc, triethyl citrate and titanium dioxide.
Each capsule is imprinted with blue pharmaceutical ink which contains butyl alcohol, dehydrated alcohol, FD & C Blue # 2 aluminum lake, isopropyl alcohol, propylene glycol, shellac, strong ammonia solution and having one circular line which contains FD & C Blue No. 2 aluminum lake, gelatin and polysorbate 80.
Questions Or Comments?
Call toll-free 1-8 77 - 835 - 5472
Mon – Fri 9AM – 5PM EST.
Made in IndiaFor most recent product information, visit www.amneal.com
Otc - Purpose Section
Principal Display Panel
NDC 69238-1050-4
Esomeprazole Magnesium Delayed-Release Capsules, USP/Acid Reducer (OTC)
20 mg
14 Capsules (Container Label)
Amneal Pharmaceuticals LLC
 Â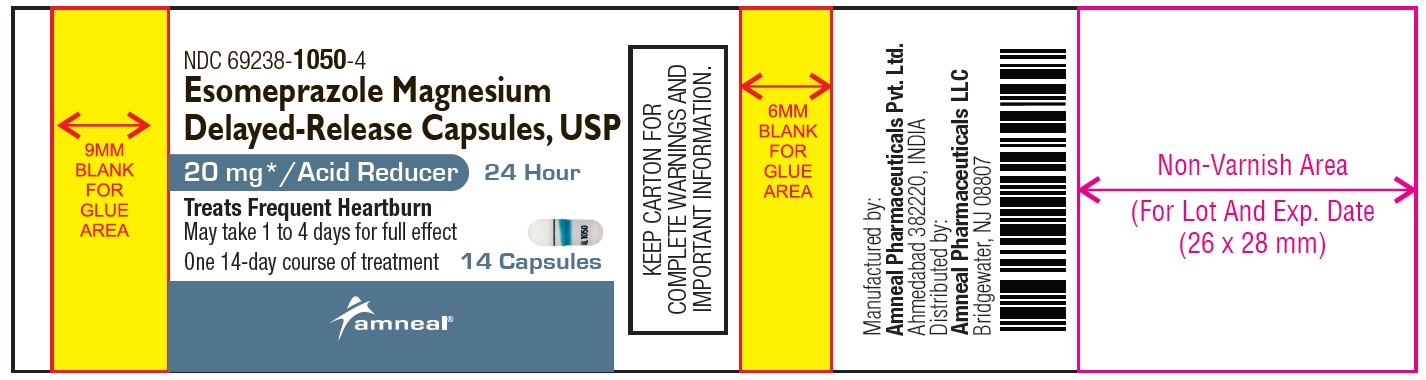




NDC 69238-1050-3
Esomeprazole Magnesium Delayed-Release Capsules, USP /Acid Reducer (OTC)
20 mg
42 Capsules (Carton Pack)
Amneal Pharmaceuticals LLC

DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site