Oxycodone Hydrochloride 30 MG Oral Tablet
INDICATIONS AND USAGE Oxycodone hydrochloride tablets are an immediate-release oral formulation of oxycodone hydrochloride indicated for the management of moderate to severe pain where the use of an opioid analgesic is appropriate.
major pharmaceuticals
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site




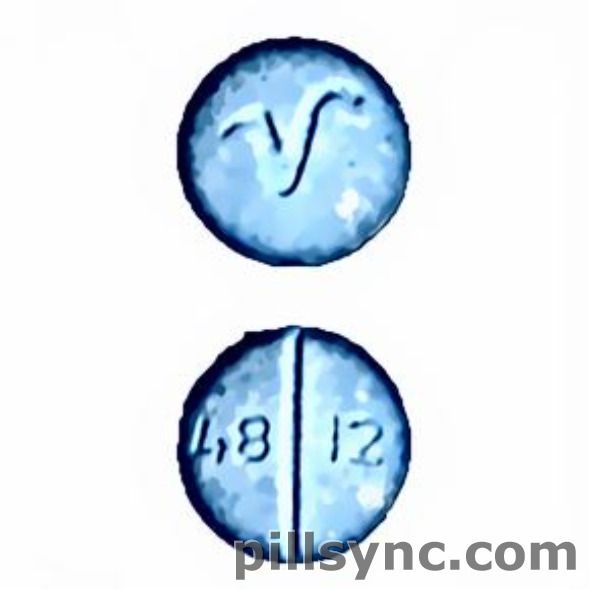
HOW SUPPLIED Oxycodone Hydrochloride Tablets, USP 10 mg: pink, round, convex, scored tablets, debossed “4993” on one side and debossed “V” on the reverse side, are supplied as follows: Carton of 100 tablets (10 tablets each blister pack x 10) NDC 0904-6590-61 30 mg: light blue, round, convex, scored tablets, debossed “4812” on one side and debossed “V” on the reverse side, are supplied as follows: Carton of 100 tablets (10 tablets each blister pack x 10) NDC 0904-6591-61 DEA Order Form Required
More pills like ROUND V 48 12