metoprolol succinate metoprolol succinate er tablets tablet film coated extended release
1 INDICATIONS AND USAGE Metoprolol succinate, is a beta-adrenergic blocker indicated for the treatment of: • Hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. ( 1.1 ) • Angina Pectoris. ( 1.2 ) • Heart Failure, to reduce the risk of cardiovascular mortality and heart failure hospitalizations in patients with heart failure ( 1.3 ) 1.1 Hypertension Metoprolol succinate extended-release tablets are indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure lowers the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes including metoprolol. Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than 1 drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program’s Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC). Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly. Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal. Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy. Metoprolol succinate extended-release tablets may be administered with other antihypertensive agents. 1.2 Angina Pectoris Metoprolol succinate extended-release tablets are indicated in the long-term treatment of angina pectoris, to reduce angina attacks and to improve exercise tolerance. 1.3 Heart Failure Metoprolol succinate extended-release tablets are indicated to reduce the risk of cardiovascular mortality and heart-failure hospitalization in patients with heart failure.
ascend laboratories, llc
Related Pills
oxycodone and acetaminophen tablet
direct rx
oxycodone hydrochloride oxycodone tablet
amneal pharmaceuticals of new york, llc
Metronidazole 500 MG Oral Tablet
Pliva Inc.
oxycodone and acetaminophen tablet
actavis elizabeth llc
oxycodone and acetaminophen tablet
actavis elizabeth llc
DISCLAIMER:
"This tool does not provide medical advice, and is for informational and educational purposes only, and is not a substitute for professional medical advice, treatment or diagnosis. Call your doctor to receive medical advice. If you think you may have a medical emergency, please dial 911."
"Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. We may limit or otherwise restrict your access to the API in line with our Terms of Service."
"This product uses publicly available data from the U.S. National Library of Medicine (NLM), National Institutes of Health, Department of Health and Human Services; NLM is not responsible for the product and does not endorse or recommend this or any other product."
PillSync may earn a commission via links on our site




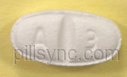






16 HOW SUPPLIED/STORAGE AND HANDLING Metoprolol Succinate Extended-Release Tablets, USP are available containing 23.75 mg, 47.5 mg, 95 mg or 190 mg of metoprolol succinate, USP equivalent to 25 mg, 50 mg, 100 mg or 200 mg of metoprolol tartrate, USP, respectively. 25 mg tablets: white to off white, oval shape, biconvex film coated tablet with breakline on one side and debossed with 'A' and '3' on each side of breakline on other side. They are available as follows: 67877-590-30 Bottles of 30 tablets 67877-590-60 Bottles of 60 tablets 67877-590-01 Bottles of 100 tablets 67877-590-05 Bottles of 500 tablets 67877-590-10Bottles of 1000 tablets 50 mg tablets: white to off white, round shape, biconvex film coated tablet with breakline on one side and debossed with 'A50' on other side. They are available as follows: 67877-591-30 Bottles of 30 tablets 67877-591-60 Bottles of 60 tablets 67877-591-01 Bottles of 100 tablets 67877-591-05 Bottles of 500 tablets 67877-591-10 Bottles of 1000 tablets 100 mg tablets: white to off white, round shape, biconvex film coated tablet with breakline on one side and debossed with 'A100' on other side. They are available as follows: 67877-592-30 Bottles of 30 tablets 67877-592-60 Bottles of 60 tablets 67877-592-01 Bottles of 100 tablets 67877-592-05 Bottles of 500 tablets 67877-592-10 Bottles of 1000 tablets 200 mg tablets: white to off white, oval shape, biconvex film coated tablet with breakline on one side and debossed with 'A200' on other side. They are available as follows: 67877-593-30 Bottles of 30 tablets 67877-593-60 Bottles of 60 tablets 67877-593-01 Bottles of 100 tablets 67877-593-05 Bottles of 500 tablets 67877-593-10 Bottles of 1000 tablets Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
More pills like OVAL A 3